Related Posts
BLOG HOME
- Subscription Billing Auto Forecast for SaaS Companies using Dynamics 365 Business Central
- Top 50 Opportunities Report for SaaS Companies using Dynamics 365 BC
- Subscription Sales by Contract Length Report for SaaS Companies using Dynamics 365 Business Central
- Multi-Year Profit & Loss Forecast Model for SaaS Companies using Dynamics 365 Business Central
- Lead Target Model for SaaS Companies using Dynamics 365 Business Central
Clinical Trials Analysis Report for Pharmaceutical Companies
What is
a
Clinical Trials Analysis Report
? Clinical Trials Reports are considered operational monitoring tools and are used by product and trial managers to analyze expenses and statistical KPIs related to their drug trial programs. Some of the main functionality in this type of graphical report is that it offers comparative analysis for four different metrics, including: 1) Target versus actual cost by drug trial, 2) Target versus actual hours by drug trial, 3) Target versus actual participants by drug trial, and 4) Target versus actual cost by status. Below the charts (not visible in the image) is a report section with figures. You find an example of this type of graphical report below.
Purpose of
Clinical Trials Analysis Reports Pharmaceutical companies use Clinical Trials Analysis Reports to give managers an easy and graphical way to keep an eye on essential product trial metrics. When used as part of good business practices in Product and Clinical Trials departments, a company can improve its program planning, and it can reduce the chances that there are cost overruns or issues with trial participation.
Example of a
Clinical Trials Analysis Report Here is an example of a Clinical Trials Analysis Report with cost, hours and participant analysis. [caption id="" align="alignnone" width="2560"]
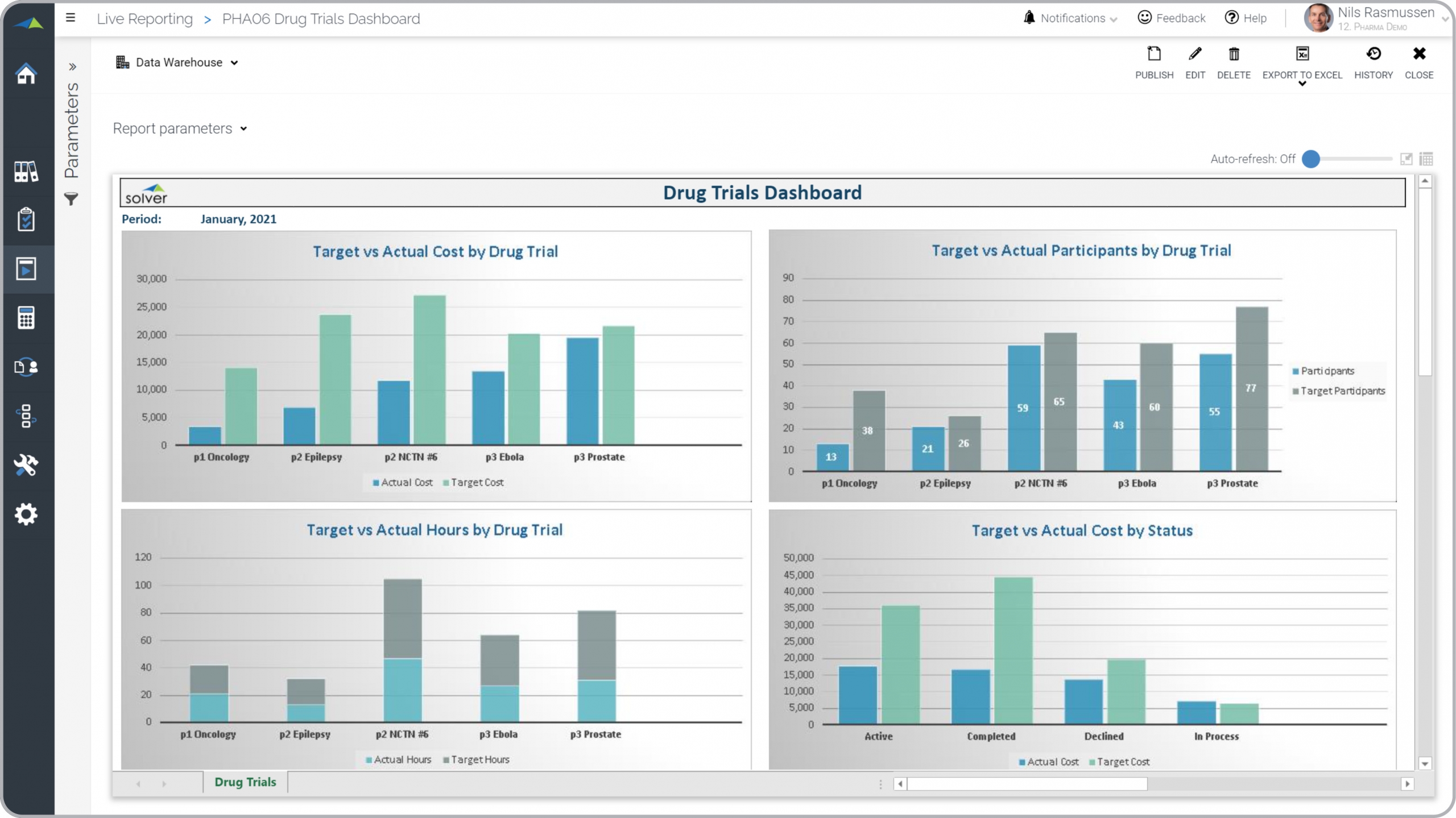 Example of a Clinical Trials Analysis Report for Pharmaceutical Companies[/caption] You can find hundreds of additional examples
here
Who Uses This Type of
Graphical report
? The typical users of this type of graphical report are: Program managers, R&D managers, controllers, analysts, executives.
Other Reports Often Used in Conjunction with
Clinical Trials Analysis Reports Progressive Product and Clinical Trials departments sometimes use several different Clinical Trials Analysis Reports, along with clinical trial KPI dashboards, clinical trial reports, profit & loss reports, budget models and other management and control tools.
Where Does the Data for Analysis Originate From? The Actual (historical transactions) data typically comes from clinical trial software and enterprise resource planning (ERP) systems like: Microsoft Dynamics 365 (D365) Finance, Microsoft Dynamics 365 Business Central (D365 BC), Microsoft Dynamics AX, Microsoft Dynamics NAV, Microsoft Dynamics GP, Microsoft Dynamics SL, Sage Intacct, Sage 100, Sage 300, Sage 500, Sage X3, SAP Business One, SAP ByDesign, Acumatica, Netsuite and others. In analyses where budgets or forecasts are used, the planning data most often originates from in-house Excel spreadsheet models or from professional corporate performance management (CPM/EPM) solutions.
What Tools are Typically used for Reporting, Planning and Dashboards? Examples of business software used with the data and ERPs mentioned above are:
Example of a Clinical Trials Analysis Report for Pharmaceutical Companies[/caption] You can find hundreds of additional examples
here
Who Uses This Type of
Graphical report
? The typical users of this type of graphical report are: Program managers, R&D managers, controllers, analysts, executives.
Other Reports Often Used in Conjunction with
Clinical Trials Analysis Reports Progressive Product and Clinical Trials departments sometimes use several different Clinical Trials Analysis Reports, along with clinical trial KPI dashboards, clinical trial reports, profit & loss reports, budget models and other management and control tools.
Where Does the Data for Analysis Originate From? The Actual (historical transactions) data typically comes from clinical trial software and enterprise resource planning (ERP) systems like: Microsoft Dynamics 365 (D365) Finance, Microsoft Dynamics 365 Business Central (D365 BC), Microsoft Dynamics AX, Microsoft Dynamics NAV, Microsoft Dynamics GP, Microsoft Dynamics SL, Sage Intacct, Sage 100, Sage 300, Sage 500, Sage X3, SAP Business One, SAP ByDesign, Acumatica, Netsuite and others. In analyses where budgets or forecasts are used, the planning data most often originates from in-house Excel spreadsheet models or from professional corporate performance management (CPM/EPM) solutions.
What Tools are Typically used for Reporting, Planning and Dashboards? Examples of business software used with the data and ERPs mentioned above are:
 Example of a Clinical Trials Analysis Report for Pharmaceutical Companies[/caption] You can find hundreds of additional examples
here
Who Uses This Type of
Graphical report
? The typical users of this type of graphical report are: Program managers, R&D managers, controllers, analysts, executives.
Other Reports Often Used in Conjunction with
Clinical Trials Analysis Reports Progressive Product and Clinical Trials departments sometimes use several different Clinical Trials Analysis Reports, along with clinical trial KPI dashboards, clinical trial reports, profit & loss reports, budget models and other management and control tools.
Where Does the Data for Analysis Originate From? The Actual (historical transactions) data typically comes from clinical trial software and enterprise resource planning (ERP) systems like: Microsoft Dynamics 365 (D365) Finance, Microsoft Dynamics 365 Business Central (D365 BC), Microsoft Dynamics AX, Microsoft Dynamics NAV, Microsoft Dynamics GP, Microsoft Dynamics SL, Sage Intacct, Sage 100, Sage 300, Sage 500, Sage X3, SAP Business One, SAP ByDesign, Acumatica, Netsuite and others. In analyses where budgets or forecasts are used, the planning data most often originates from in-house Excel spreadsheet models or from professional corporate performance management (CPM/EPM) solutions.
What Tools are Typically used for Reporting, Planning and Dashboards? Examples of business software used with the data and ERPs mentioned above are:
Example of a Clinical Trials Analysis Report for Pharmaceutical Companies[/caption] You can find hundreds of additional examples
here
Who Uses This Type of
Graphical report
? The typical users of this type of graphical report are: Program managers, R&D managers, controllers, analysts, executives.
Other Reports Often Used in Conjunction with
Clinical Trials Analysis Reports Progressive Product and Clinical Trials departments sometimes use several different Clinical Trials Analysis Reports, along with clinical trial KPI dashboards, clinical trial reports, profit & loss reports, budget models and other management and control tools.
Where Does the Data for Analysis Originate From? The Actual (historical transactions) data typically comes from clinical trial software and enterprise resource planning (ERP) systems like: Microsoft Dynamics 365 (D365) Finance, Microsoft Dynamics 365 Business Central (D365 BC), Microsoft Dynamics AX, Microsoft Dynamics NAV, Microsoft Dynamics GP, Microsoft Dynamics SL, Sage Intacct, Sage 100, Sage 300, Sage 500, Sage X3, SAP Business One, SAP ByDesign, Acumatica, Netsuite and others. In analyses where budgets or forecasts are used, the planning data most often originates from in-house Excel spreadsheet models or from professional corporate performance management (CPM/EPM) solutions.
What Tools are Typically used for Reporting, Planning and Dashboards? Examples of business software used with the data and ERPs mentioned above are:
- Native ERP report writers and query tools
- Spreadsheets (for example Microsoft Excel)
- Corporate Performance Management (CPM) tools (for example Solver)
- Dashboards (for example Microsoft Power BI and Tableau)
- View 100’s of reporting, consolidations, planning, budgeting, forecasting and dashboard examples here
- View a Pharma Industry white paper and other industry-specific information here
- See how reports are designed in a modern report writer using a cloud-connected Excel add-in writer
- Discover how the Solver CPM solution delivers financial and operational reporting
- Discover how the Solver CPM solution delivers planning, budgeting and forecasting
- Watch demo videos of reporting, planning and dashboards
TAGS: Reporting, Planning, Forecasting, Budgeting, CPM, KPIs, ERP, Dashboards, Industry, Financial Reporting, Template Library
Global Headquarters
Solver, Inc.
Phone: +1 (310) 691-5300
